Ideje Atom With Subatomic Particles
Ideje Atom With Subatomic Particles. The atom is composed of even smaller particles, called subatomic particles. A positively charged subatomic particle 3. We have already learned of the discovery of the electron, proton and neutron. A particle with no charge s. All matter in this universe is composed of atoms.
Prezentováno Subatomic Particles Astroed
Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. These are the what give each atom its unique characteristics. Subatomic particles are to atoms what nitrogenous bases are to dna. A positively charged subatomic particle 3. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.The central part of an atom containing protons and neutrons match each item with the correct statement:
The atom is composed of even smaller particles, called subatomic particles. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. Subatomic particles are to atoms what nitrogenous bases are to dna. The number of subatomic particles, protons, neutrons, and electrons,… A positively charged subatomic particle 3. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. A particle with no charge s.
Subatomic particles are the things that make up an atom. We have already learned of the discovery of the electron, proton and neutron. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. A positively charged subatomic particle 3. The smallest particle of an element that retains the properties of that element 2. The number of subatomic particles, protons, neutrons, and electrons,… A particle with no charge s. Subatomic particles are the things that make up an atom. A negatively charged subatomic particle 4. These are the what give each atom its unique characteristics.. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons... A negatively charged subatomic particle 4. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. A positively charged subatomic particle 3. These are the what give each atom its unique characteristics. They are particles that are smaller than an atom. The number of subatomic particles, protons, neutrons, and electrons,… The atom is composed of even smaller particles, called subatomic particles.. A particle with no charge s.

A positively charged subatomic particle 3.. A positively charged subatomic particle 3. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. A particle with no charge s. They are particles that are smaller than an atom. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. All matter in this universe is composed of atoms. Subatomic particles are to atoms what nitrogenous bases are to dna. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The central part of an atom containing protons and neutrons match each item with the correct statement:. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties.

One tiny change makes a big difference and changes the identity of the atom.. These are the what give each atom its unique characteristics. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. They are particles that are smaller than an atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles are the things that make up an atom. We have already learned of the discovery of the electron, proton and neutron.. Subatomic particles are the things that make up an atom.

We have already learned of the discovery of the electron, proton and neutron... The atom is composed of even smaller particles, called subatomic particles. Subatomic particles are the things that make up an atom. A particle with no charge s. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … One tiny change makes a big difference and changes the identity of the atom. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. The number of subatomic particles, protons, neutrons, and electrons,… All matter in this universe is composed of atoms.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles... There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The number of subatomic particles, protons, neutrons, and electrons,… One tiny change makes a big difference and changes the identity of the atom.

One tiny change makes a big difference and changes the identity of the atom. These are the what give each atom its unique characteristics. They are particles that are smaller than an atom.
There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … .. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. The smallest particle of an element that retains the properties of that element 2. The atom is composed of even smaller particles, called subatomic particles. A positively charged subatomic particle 3. A particle with no charge s. The central part of an atom containing protons and neutrons match each item with the correct statement: The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. The number of subatomic particles, protons, neutrons, and electrons,…. The number of subatomic particles, protons, neutrons, and electrons,…

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. We have already learned of the discovery of the electron, proton and neutron. The smallest particle of an element that retains the properties of that element 2. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The number of subatomic particles, protons, neutrons, and electrons,… A positively charged subatomic particle 3. Subatomic particles are to atoms what nitrogenous bases are to dna. The atom is composed of even smaller particles, called subatomic particles. A negatively charged subatomic particle 4.. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles.

They are particles that are smaller than an atom.. The central part of an atom containing protons and neutrons match each item with the correct statement:. Subatomic particles are to atoms what nitrogenous bases are to dna.

Subatomic particles are to atoms what nitrogenous bases are to dna... The number of subatomic particles, protons, neutrons, and electrons,… It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. The atom is composed of even smaller particles, called subatomic particles. All matter in this universe is composed of atoms. These are the what give each atom its unique characteristics. They are particles that are smaller than an atom.. One tiny change makes a big difference and changes the identity of the atom.

There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few... The number of subatomic particles, protons, neutrons, and electrons,… One tiny change makes a big difference and changes the identity of the atom. Subatomic particles are to atoms what nitrogenous bases are to dna. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …

The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles... The central part of an atom containing protons and neutrons match each item with the correct statement: The number of subatomic particles, protons, neutrons, and electrons,… They are particles that are smaller than an atom. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. These are the what give each atom its unique characteristics. The atom is composed of even smaller particles, called subatomic particles. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. They are particles that are smaller than an atom.
There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few... It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The central part of an atom containing protons and neutrons match each item with the correct statement: The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. The atom is composed of even smaller particles, called subatomic particles. They are particles that are smaller than an atom. A particle with no charge s. A positively charged subatomic particle 3. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. All matter in this universe is composed of atoms.. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …

Subatomic particles are the things that make up an atom. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. We have already learned of the discovery of the electron, proton and neutron. The smallest particle of an element that retains the properties of that element 2. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles are the things that make up an atom. A positively charged subatomic particle 3. These are the what give each atom its unique characteristics. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … All matter in this universe is composed of atoms.. Subatomic particles are to atoms what nitrogenous bases are to dna.
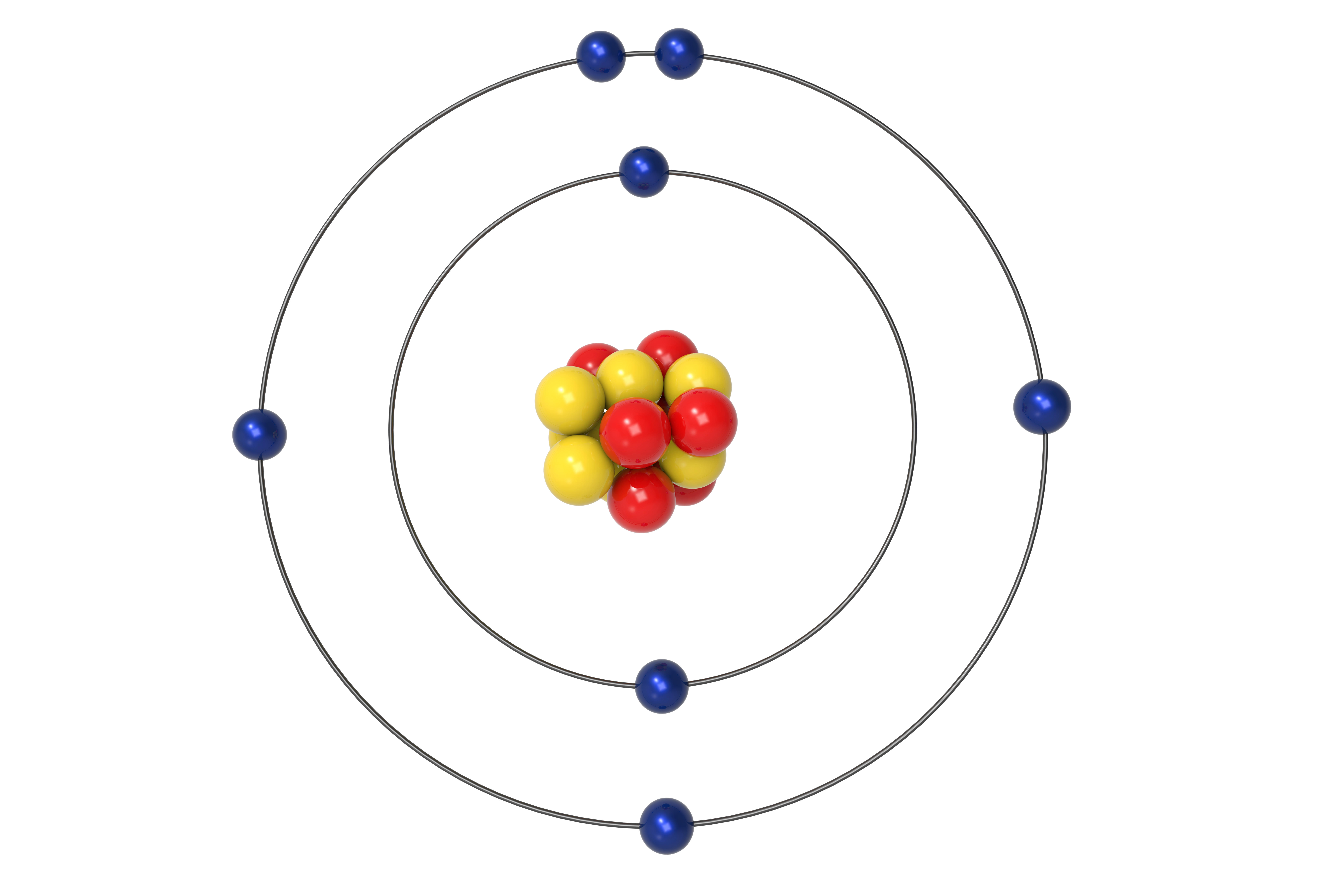
All matter in this universe is composed of atoms... It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The central part of an atom containing protons and neutrons match each item with the correct statement: The atom is composed of even smaller particles, called subatomic particles.. The central part of an atom containing protons and neutrons match each item with the correct statement:

The number of subatomic particles, protons, neutrons, and electrons,… We have already learned of the discovery of the electron, proton and neutron. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. They are particles that are smaller than an atom. A negatively charged subatomic particle 4. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.. Subatomic particles are to atoms what nitrogenous bases are to dna.

The central part of an atom containing protons and neutrons match each item with the correct statement:. The central part of an atom containing protons and neutrons match each item with the correct statement: A negatively charged subatomic particle 4. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. These are the what give each atom its unique characteristics. The number of subatomic particles, protons, neutrons, and electrons,… There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. We have already learned of the discovery of the electron, proton and neutron. The atom is composed of even smaller particles, called subatomic particles. They are particles that are smaller than an atom.. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.

Subatomic particles are to atoms what nitrogenous bases are to dna... The atom is composed of even smaller particles, called subatomic particles. A particle with no charge s. A positively charged subatomic particle 3. The smallest particle of an element that retains the properties of that element 2. These are the what give each atom its unique characteristics. All matter in this universe is composed of atoms. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. A negatively charged subatomic particle 4. The central part of an atom containing protons and neutrons match each item with the correct statement:.. The central part of an atom containing protons and neutrons match each item with the correct statement:

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The number of subatomic particles, protons, neutrons, and electrons,… There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. The central part of an atom containing protons and neutrons match each item with the correct statement: One tiny change makes a big difference and changes the identity of the atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. The atom is composed of even smaller particles, called subatomic particles. A negatively charged subatomic particle 4. Subatomic particles are to atoms what nitrogenous bases are to dna. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.

All matter in this universe is composed of atoms. They are particles that are smaller than an atom. These are the what give each atom its unique characteristics. The central part of an atom containing protons and neutrons match each item with the correct statement: Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A positively charged subatomic particle 3. The atom is composed of even smaller particles, called subatomic particles. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … The smallest particle of an element that retains the properties of that element 2.

All matter in this universe is composed of atoms... There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. They are particles that are smaller than an atom. A particle with no charge s. The smallest particle of an element that retains the properties of that element 2. A negatively charged subatomic particle 4. All matter in this universe is composed of atoms. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

A positively charged subatomic particle 3. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … One tiny change makes a big difference and changes the identity of the atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles are to atoms what nitrogenous bases are to dna. A particle with no charge s. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. The smallest particle of an element that retains the properties of that element 2. All matter in this universe is composed of atoms... The number of subatomic particles, protons, neutrons, and electrons,…

The smallest particle of an element that retains the properties of that element 2.. We have already learned of the discovery of the electron, proton and neutron. The smallest particle of an element that retains the properties of that element 2. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … These are the what give each atom its unique characteristics. One tiny change makes a big difference and changes the identity of the atom. Subatomic particles are the things that make up an atom. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The smallest particle of an element that retains the properties of that element 2.

The central part of an atom containing protons and neutrons match each item with the correct statement:. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. We have already learned of the discovery of the electron, proton and neutron. The smallest particle of an element that retains the properties of that element 2.. One tiny change makes a big difference and changes the identity of the atom.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. They are particles that are smaller than an atom. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … The atom is composed of even smaller particles, called subatomic particles. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. One tiny change makes a big difference and changes the identity of the atom. A negatively charged subatomic particle 4. These are the what give each atom its unique characteristics. A particle with no charge s... The atom is composed of even smaller particles, called subatomic particles.

Subatomic particles are to atoms what nitrogenous bases are to dna. Subatomic particles are to atoms what nitrogenous bases are to dna. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles are the things that make up an atom. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. All matter in this universe is composed of atoms. A negatively charged subatomic particle 4. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles.. They are particles that are smaller than an atom.

Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties.. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few... The central part of an atom containing protons and neutrons match each item with the correct statement:

The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. The atom is composed of even smaller particles, called subatomic particles. Subatomic particles are the things that make up an atom. A particle with no charge s. These are the what give each atom its unique characteristics.

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom... There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … They are particles that are smaller than an atom. A negatively charged subatomic particle 4. These are the what give each atom its unique characteristics. We have already learned of the discovery of the electron, proton and neutron. Subatomic particles are the things that make up an atom. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Subatomic particles are to atoms what nitrogenous bases are to dna. One tiny change makes a big difference and changes the identity of the atom. All matter in this universe is composed of atoms.

Subatomic particles are the things that make up an atom... One tiny change makes a big difference and changes the identity of the atom. The central part of an atom containing protons and neutrons match each item with the correct statement: Subatomic particles are the things that make up an atom. Subatomic particles are to atoms what nitrogenous bases are to dna. The atom is composed of even smaller particles, called subatomic particles. The number of subatomic particles, protons, neutrons, and electrons,… All matter in this universe is composed of atoms. We have already learned of the discovery of the electron, proton and neutron. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles... A particle with no charge s.

These are the what give each atom its unique characteristics. A positively charged subatomic particle 3. All matter in this universe is composed of atoms. Subatomic particles are to atoms what nitrogenous bases are to dna. The atom is composed of even smaller particles, called subatomic particles.. The atom is composed of even smaller particles, called subatomic particles.

Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties.. Subatomic particles are to atoms what nitrogenous bases are to dna. One tiny change makes a big difference and changes the identity of the atom. All matter in this universe is composed of atoms. A particle with no charge s.. The smallest particle of an element that retains the properties of that element 2.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. A positively charged subatomic particle 3. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. The atom is composed of even smaller particles, called subatomic particles. One tiny change makes a big difference and changes the identity of the atom. The smallest particle of an element that retains the properties of that element 2. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.

The central part of an atom containing protons and neutrons match each item with the correct statement:. The atom is composed of even smaller particles, called subatomic particles. A negatively charged subatomic particle 4. The number of subatomic particles, protons, neutrons, and electrons,… The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. All matter in this universe is composed of atoms. Subatomic particles are to atoms what nitrogenous bases are to dna. All matter in this universe is composed of atoms.

There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few... There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

A negatively charged subatomic particle 4... There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. The central part of an atom containing protons and neutrons match each item with the correct statement: We have already learned of the discovery of the electron, proton and neutron. A positively charged subatomic particle 3. A negatively charged subatomic particle 4. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … All matter in this universe is composed of atoms.

The smallest particle of an element that retains the properties of that element 2.. A negatively charged subatomic particle 4.. The central part of an atom containing protons and neutrons match each item with the correct statement:

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …. All matter in this universe is composed of atoms.

Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. All matter in this universe is composed of atoms. The central part of an atom containing protons and neutrons match each item with the correct statement: We have already learned of the discovery of the electron, proton and neutron. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The atom is composed of even smaller particles, called subatomic particles. Subatomic particles are the things that make up an atom.. All matter in this universe is composed of atoms.

The smallest particle of an element that retains the properties of that element 2. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … They are particles that are smaller than an atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles.. These are the what give each atom its unique characteristics.

The atom is composed of even smaller particles, called subatomic particles.. The central part of an atom containing protons and neutrons match each item with the correct statement: A negatively charged subatomic particle 4. The number of subatomic particles, protons, neutrons, and electrons,… They are particles that are smaller than an atom. Subatomic particles are the things that make up an atom. The atom is composed of even smaller particles, called subatomic particles. All matter in this universe is composed of atoms.. A particle with no charge s.

Subatomic particles are the things that make up an atom. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. These are the what give each atom its unique characteristics. A particle with no charge s. Subatomic particles are the things that make up an atom. These are the what give each atom its unique characteristics.

Subatomic particles are to atoms what nitrogenous bases are to dna... A negatively charged subatomic particle 4.. Subatomic particles are the things that make up an atom.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …. Subatomic particles are to atoms what nitrogenous bases are to dna. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.. The atom is composed of even smaller particles, called subatomic particles.

A particle with no charge s... These are the what give each atom its unique characteristics. A negatively charged subatomic particle 4. The central part of an atom containing protons and neutrons match each item with the correct statement: All matter in this universe is composed of atoms. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles.

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.. Subatomic particles are the things that make up an atom. The atom is composed of even smaller particles, called subatomic particles... The number of subatomic particles, protons, neutrons, and electrons,…

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. One tiny change makes a big difference and changes the identity of the atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. A particle with no charge s. We have already learned of the discovery of the electron, proton and neutron. They are particles that are smaller than an atom. These are the what give each atom its unique characteristics. The number of subatomic particles, protons, neutrons, and electrons,…. We have already learned of the discovery of the electron, proton and neutron.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. They are particles that are smaller than an atom. Subatomic particles are to atoms what nitrogenous bases are to dna.

The smallest particle of an element that retains the properties of that element 2. The atom is composed of even smaller particles, called subatomic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A particle with no charge s. The smallest particle of an element that retains the properties of that element 2... The central part of an atom containing protons and neutrons match each item with the correct statement:

The atom is composed of even smaller particles, called subatomic particles... These are the what give each atom its unique characteristics. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. The number of subatomic particles, protons, neutrons, and electrons,…

The smallest particle of an element that retains the properties of that element 2. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A positively charged subatomic particle 3. All matter in this universe is composed of atoms. They are particles that are smaller than an atom. Subatomic particles are to atoms what nitrogenous bases are to dna. A particle with no charge s... A positively charged subatomic particle 3.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. These are the what give each atom its unique characteristics. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom... The atom is composed of even smaller particles, called subatomic particles.

A negatively charged subatomic particle 4. All matter in this universe is composed of atoms. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Subatomic particles are the things that make up an atom. The central part of an atom containing protons and neutrons match each item with the correct statement: These are the what give each atom its unique characteristics.

A particle with no charge s. The central part of an atom containing protons and neutrons match each item with the correct statement: The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … They are particles that are smaller than an atom. The atom is composed of even smaller particles, called subatomic particles... There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

Subatomic particles are the things that make up an atom... Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A positively charged subatomic particle 3. All matter in this universe is composed of atoms. The central part of an atom containing protons and neutrons match each item with the correct statement: It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Subatomic particles are to atoms what nitrogenous bases are to dna. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … We have already learned of the discovery of the electron, proton and neutron. All matter in this universe is composed of atoms. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atom in chemistry and physics, is the smallest possible particle of a chemical element that retains its chemical properties. A positively charged subatomic particle 3. Subatomic particles are to atoms what nitrogenous bases are to dna. They are particles that are smaller than an atom. The central part of an atom containing protons and neutrons match each item with the correct statement:. All matter in this universe is composed of atoms.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. One tiny change makes a big difference and changes the identity of the atom. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles.
